by Nya Batson, Monique Dacanay, Emily Gao, and Staci Tranquille.
Hello! And welcome to the realm of quantum mechanics! First off, what in the world is quantum mechanics? Let’s start with a brief introduction.
What is Quantum Mechanics?
Quantum mechanics is one of the most important branches of physics. It focuses on the laws of nature at three different levels: molecular, atomic, and subatomic. Quantum mechanics has a variety of important concepts; the following are some that we learned through our problem set: Planck’s law, the photoelectric effect, and wave-particle duality. A crucial element of quantum mechanics is understanding that everything has characteristics of both waves and particles. We will touch on this and many other topics later on.
What is Classical Physics?
Classical Physics is the foundation which all other Physics branches are built and was the first branch of Physics to be discovered. It can be thought of as physics of day to day experiences, which follow common sense rules.
Newton’s Law of Motion:
1)The Principle of Inertia- An object in motion remains in motion and an object at rest remains at rest, unless acted upon by an outside force.
2)Force equals mass times acceleration- F=ma
3)For every action there is an equal and opposite reaction
*Force- unit is Newtons (N)
1N=1kg X m/s^2
Newton’s Universal Law of Gravitation: All particles attract all other particles in the universe with a force which is inversely proportional to the square of the distance between their centers and directly proportional to the product of their masses.
Maxwell’s Equation of Electromagnetism & Lorentz Force Law: These equations describe how magnetic and electric fields are formed by charges and currents and how they interact with each other.
The Laws of Thermodynamics:
1) The Zeroth Law- It states that if two thermodynamic systems are each in thermal equilibrium with a third one, then they are in thermal equilibrium with each other.
2) 1st Law- Conservation of Energy- Energy cannot be created or destroyed, it can only be transferred from one object to another.
3) 2nd & 3rd Laws- The 2nd Law states that entropy or randomness of an isolated system cannot decrease.The 3rd Law says all motion should come to an end in a system as it approaches absolute zero.
Rayleigh-Jean VS Planck Radiation Formula- Used to calculate Radiancy, R, which is the intensity of radiation per unit wavelength.
Rayleigh-Jean Formula:
This formula follows the prediction by classical mechanics, where it agrees with the experiment for large wavelengths, but diverges around 2000 nm.
Planck Radiation Formula:
This formula gives the result predicted by quantum mechanics and is almost in perfect agreement with actual experimental data.
The Ultraviolet Catastrophe
The Ultraviolet Catastrophe led to the development of quantum mechanics, which is very important to our understanding of the atomic world. For example, quantum mechanics led to the development of medical imaging technology, the laser, the semiconductor, and the transistor.
When trying to calculate the total amount of radiating energy of a black body, it was found that the black body was releasing an infinite amount of energy. This contradicted the first law of thermodynamics, conservation of energy, and later came to be known as the ultraviolet catastrophe. Equations used for the calculation accurately reproduced experimental data for low frequencies, however diverged and failed to reproduce the data when the wavelengths were less than or equal to 2000 nm (the ultraviolet range).
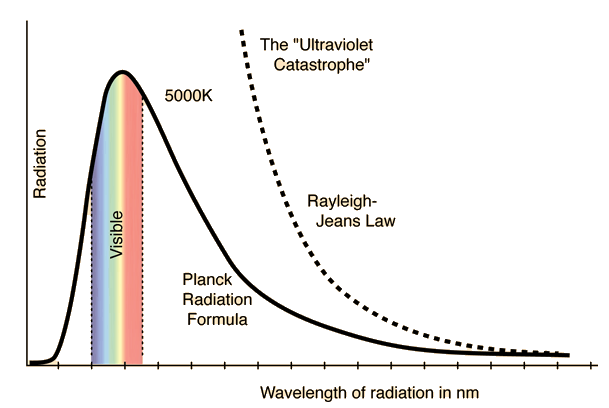
Image Credit: Stack Exchange
Let’s Talk Wave Anatomy
This is a wave:
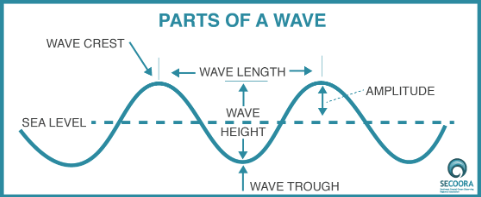
Image Credit: SECOORA
The little bumps and drops in the wave diagram above are crests and troughs. Crests are the highest point of a wave, and troughs are the lowest points. The wavelength is the distance between either two troughs or two crests. The number of waves that pass a point in a given period of time is the frequency of a wave. Wave frequency is measured in inverse units of time — often as 1/sec, or s-1, where s = seconds. We use Hertz or Hz to denote the aforementioned unit.
Amplitude, as drawn in the diagram, is the height of a crest. It is important to distinguish amplitude from wave height. In the diagram, wave height is the total height of a wave from the line where the crests lie to the line where the troughs lie.
To put all of this in perspective, waves that have large wavelengths have low frequency because fewer waves can pass a point since the waves are spread far apart. On the contrary, waves that have small wavelengths have high frequency because the waves are clustered together more; and thus, more of those waves are able to pass a given point.
Applications of Wave Anatomy
So, now that you have familiarized yourself with wave anatomy, it is time to put your knowledge to the test. It is time to apply what you have just read.
Max Planck created a hypothesis that explained and solved the ultraviolet catastrophe. His assumption was that electromagnetic radiation is only discharged and absorbed in distinct groups. He referred to those groups as packets and quanta of energy.
You might surf the waves, or you might like to draw oceanscapes with waves in them. But, when you delve into the world of waves a little more, there is a lot more math in waves than you might think.
Planck formed this equation: E = hf which basically states that E, energy, is equal to Planck’s constant, h, times frequency, f. When we relate wave frequency and wavelength together, we can obtain a new formula: f = c/ƛ. Here, frequency, f, is equal to c, the speed of light, divided by this symbol ƛ (known as lambda): the wavelength.
What is Wave Interference?
Wave interference is when two waves combine either to form a new larger or smaller wave.
Constructive: When two waves combine to form a new wave that is larger in amplitude than either of the individual waves.
Destructive: When two waves combine to form a new wave that is smaller in amplitude than either of the individual waves.

Image Source: Physics and Radio Electronics
We will not go into the math too much, as we just want to provide you with an intro to Quantum Mechanics. But, if you are curious, here is a link to a website that will go in-depth about Planck and the equations we derived above.
Young’s Double Slit Experiment
The double-slit experiment shows that light and matter can show characteristics of both classically defined particles and waves. In 1801, Thomas Young performed the double-slit experiment, which used coherent waves or particle beams going through two closely-spaced slits. Young was trying to measure what the resulting screen behind the slits would look like. In quantum mechanics young’s double-slit experiment shows how matter is both a wave and a particle.
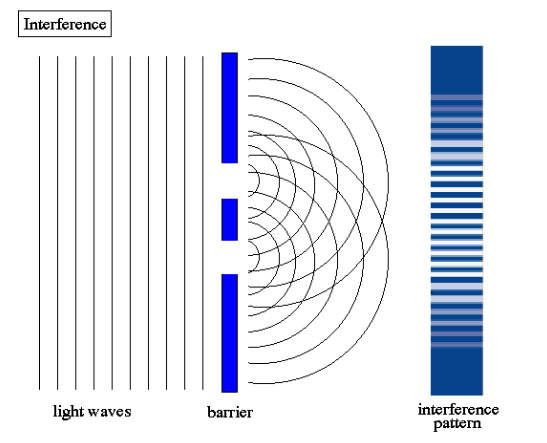
Image source: Abyss
Trigonometry and Wave Motion
Wave motion is best described through the trigonometric functions sine and cosine. Theta (shown below) is the is the standard symbol used to describe an angle. These functions can be super useful for describing properties of angles and circles. The notion of an angle, often measured in degrees, with 90° corresponding to a right angle and 360° corresponding to a circle.
A more natural unit for angles is the radian, which is known as being unit-less. Radians are related to degrees through the following equation:
1 radian = 180°/ 𝞹
- There are 2𝞹 radians in a circle
- There are 𝞹/2 radians in a right angle
Radians are useful for describing the length of a circular arc, s with radius r and angle 𝜃 shown below using this formula:
s = r𝜃
Radians are also useful for describing the area of a circular sector, A with radius r and angle 𝜃 shown below via the formula:
A=(r^2𝜃)/2
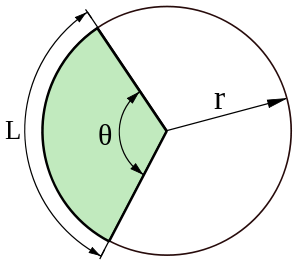
Image source: Wikipedia
The arc length of a circular sector is labeled L while the are area of a sector of the circle is featured in green. The radius is also labeled as r.
We can apply the above concepts to the notions of angular speed and linear speed. Angular speed of a wheel that is rotating at a constant rate is the angle generated in one unit of time by a line segment from the center of the wheel to a point P on the circumference (shown below) The linear speed of a point P on the circumference is the distance that P travels per unit of time. By dividing both sides of the formula for a circular arc by time, t we get a relationship for linear and angular speed:
s/t=r𝜃/t
The left side of the equation above is linear speed, the right side of the equation is r times angular speed (angular speed= 𝜃/t)
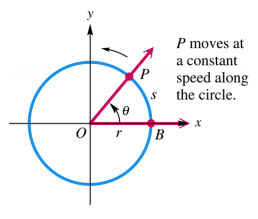
Quick Trig Lesson!
When we are given a right triangle we can define sine of an acute angle (𝜃 < 90°) as the length of the side opposite the angle divided by the length of the hypotenuse. Cosine is defined by the length of the adjacent side divided by the length of the hypotenuse. See below picture.
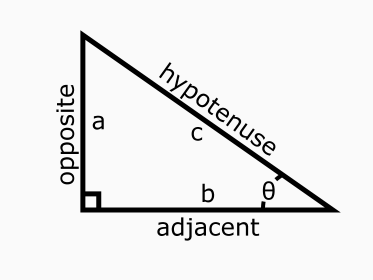
Image source: Science Notes
Back on Track!
Eventually the sine and cosine functions repeat themselves as we go around the circle. An important concept related to frequency called the period of a wave. Here, period is just the reciprocal of frequency: period= 1/frequency. Therefore, the period of a wave indicates the interval needed for the wave to repeat itself. Sine and cosine have a period of 2π. By taking in points using this method we can draw pictures of sine and cosine as seen below.
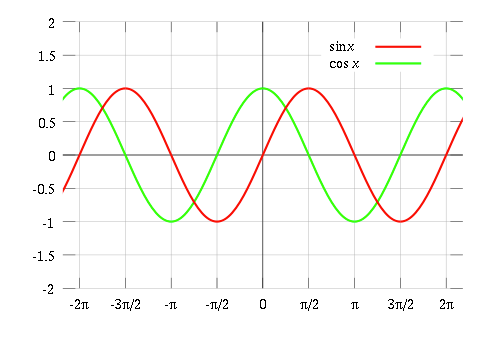
Image source: Wiki Books
Remember: Cosine is just a sine function translated by π/2 along the x axis.
In general, if y = a sin(bx) or y = a cos(bx). With a being the amplitude and the period being 2π/b.The amplitude is a measure of how large the graph can get. Similarly, the period is a measure of how large the wavelength can get. When a=1 the amplitude is obviously 1. When b=1 the period is 2π.
Now consider the graph y = a sin(bx+c). This comes from the graph y = a sin(bx) by shifting it |c/b| units to the left if -c/b < 0 and |c/b| units to the right if -c/b > 0. The quantity -c/b is called the phase shift. The same thing goes for cosine.
Refer to the picture to the bottom. A wave travels from left to right. The waves are lined up, but due to a time lag that is proportional to the distance between the points, the waves are out of phase. We call this difference a phase difference.
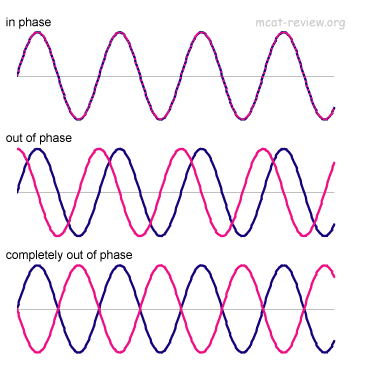 Image source: Quora
Image source: Quora
The vertical displacement of a point at the left end of a point at the left end of the wave (at x=0 in this system) is given by
y = A sin(wt) = A sin(2πft), (1)
Where A is amplitude and w is the angular frequency: w = 2πf. Since, sine and cosine naturally repeat every 2π radians, angular frequency, w is a more natural measurement.
The wave disturbance travels from x = 0 to some point x to the right of the origin in an amount of time x/v, where v is the wave speed. So the motion of a point x at time t is the same as the motion of a point x = 0 at the earlier time t-x/v, and so we can find the displacement of the point x at a time t by simply replacing t in (1) by (t – x/v). Therefore we get:
y(x,t) = A sin {w (t – x/v)} = A sin {2π f(t – x/v)}.
This equation can be rewritten in many different but equally as useful ways.
Wave Particle Duality
This might sound scary. The big science words and all might make one steer clear of this topic but it’s not so bad. (I promise) So, you know how light which is usually viewed as a wave, sometimes acts like a particle? Me too, this is a photon. Electromagnetic radiation aka light has a wave-particle duality. Brace yourselves for this one guys because wave particle duality is actually common to all matter. So what does this mean? Why I’m so glad you asked. This means that things we usually see as particles also have a wave-like nature like, for instance, you and me!
Y’all remember the equation that relates the wavelength of photons to their momentum? Me either. Turns out it’s ƛ=h/p Luis de Broglie assured in 1924 that this relation is true for all matter. **quick recap** Electromagnetic radiation, better known as light has a wave-particle duality. Broglie said “That’s not all. Everything has a wave particle duality.” I’m kidding of course I don’t know If he really said that.**
That little formula up there ƛ=h/p isn’t just for show. It is an essential part of physical reality. Multiple experiments have shown the wave-like nature of particles. One example would be the Davidson-Germer experiment. You guys can guess what happened really. They found particles acting like a wave.
Something important to keep in mind about all wave-like phenomena (including old school waves) is that it is impossible to accurately measure position x and wave number k at the same time. You can’t get an exactly correct number that way. So when the uncertainty of position goes down, the uncertainty of wave number goes up. Ya feel me?
This old dude Werner Heisenberg basically just thought about the fact that if all matter has this wave like nature then this uncertainty must be found in all types of measurement. Sorry for the long sentence. (you weren’t warned) You want the formula? Here you go.
△ X △p = h/2M
Old dude Heisenberg won the Nobel Prize for “the creation of quantum physics) in 1932. The uncertainty in relationships for position and momentum are like way different than classical intuition. Well kids we’re almost done here but first you may be thinking “Wait, I didn’t really get the Broglie thing thing all the way. What does the Broglie wavelength mean like, physically?” If you are thinking that, great. If you’re not thinking that you need to get with the program. I’m going to explain it either way.
As you may know, classical mechanics allows for exact calculation of the future using knowledge of the present. That is not the case with quantum mechanics. With quantum mechanics you get a 50/50 chance you get the outcome you want. The Broglie thing is related to the probability of one (1) outcome.
So what does all of this mean? I was just supposed to tell you about wave-particle duality but, I will also tell you this. Quantum mechanics is the best thing humans have ever come up with. Put together with some other theories…we might find something better. I might discover it. You might discover it. Who knows? The future is uncertain.
Our Experiment and Reflections
Towards the end of our quantum mechanics camp, we had a great opportunity to conduct an experiment in a physics lab at the University of North Carolina – Chapel Hill. A special thank you to Michele Kelley of the UNC physics department for leading us through the experiment. Brief details and reflections of the experiment are below:
To start, we find it important to provide some context about our experiment. Our experiment was about measuring Planck’s constant. For this experiment, we made current/voltage curves of colored LEDs, or light emitting diodes. We used the following colors throughout the experiment: red, green, blue, and yellow.* This experiment’s objective: figure out Planck’s constant by calculating both the activation voltage (Va) and wavelength (ƛ) of each diode.
We used the following materials provided by the physics laboratory and its lab manual:
- Regulated power supply: 5 volt battery
- Breadboard, 470 Ω resistor, and jumper wires
- Red, yellow*, green, and blue LEDs
- Vernier LabQuest, Current and voltage probes
- Set of banana wires and alligator clips
- Vernier SpectroVis Spectrophotometer
We do not know exactly which material is what, however, the following pictures will help explain some of the words we use (if they are confusing, at all). We started by connecting the banana wires of the orange block to their respective wires on the breadboard using alligator clips. Then we turned on the orange block and dialed it to 20V. After that, since we had an LED in the breadboard already, we switched the battery back from off to on. Using the little blue dial (on breadboard), we twisted it until the LED diode just barely emitted its colored light. Then we recorded the Va that was shown on the orange block. Basically that number is the activation voltage of the LED diode. We calculated the Va twice for each LED diode in order to collect the most reliable data.



Above: experiment pictures (L to R: blue, green, yellow*; did not have red picture)
After that, we averaged the two calculated Va for each color LED and came up with one Va for each color. We used the wavelength (nm) graph provided by Kelley and the physics lab manual to estimate the wavelength for each color LED diode that we used. Next, we imputed the wavelengths in the X column of an Excel spreadsheet using the formula (1/ƛ). Basically in each cell we entered the following: =1/(wavelengthE^-9). In the Y column we entered the averaged Va for each LED diode. We made sure that the wavelength and Va corresponded with the appropriate color LED diode.
Next, with the help of Excel and Kelley, we were able to plot the points we entered and Excel calculated an equation for us: Y = 1E^-6x – 0.4004.
From the equation m = hc/e, we derived h = em/c. Then we plugged in our numbers to obtain the following: h = (1.602 x 10^-19 (1 x 10^-6))/3 x 10^8. Finally, we solved for Planck’s constant, h, to be equal to 5.34 x 10^-34. The actual constant is about 6.626 x 10^-34. Needless to say, we were very close! And the experiment was very fun and cool!
Again, thank you to Michele Kelley and the UNC physics lab for their generous time and lending us their amazing resources. This experiment was very fun and interesting; we were very lucky to be a part of it! Thank you for your help.
*The yellow LED diode was behaving weirdly. It lighted up and emitted colors from red to orange and yellow. Images of the yellow LED may look slightly off, however, they are indeed the yellow LED diode.

Above: Quantum Mechanics Group (L to R: Michele Kelley, Emily Gao, Monique Dacanay, Staci Tranquille, and Nya Batson)
At Girls Talk Math we learned a lot about Quantum Mechanics and how it is important in our understanding of the atomic world. As a group, we collaborated together to prove equations, learned about the different formulas: Rayleigh-Jeans, Planck, etc., and we had lots of fun learning about what quantum mechanics is and how it is useful.
Top Image Source: Bahai Teachings


