By Kathryn Benedict, Olivia Fugikawa, Denna Huang, and Eleanor McAdon
Intro
Quantum mechanics is a subfield of physics. Like with any other major area of study, physics is divided into many smaller categories. Classical physics is the main one, which includes Newton’s Laws of Motion and basic principles of mechanics, like inertia and friction. Things get weird when you delve into modern physics, which includes special relativity, general relativity, and quantum mechanics. Special relativity deals with particles moving at the speed of light, general relativity works with incredibly massive objects and quantum mechanics is the physics of subatomic particles. This is what we worked on for the past two weeks and what our blog post is about!
There are many different topics that we learned about in quantum mechanics, including wave-particle duality, uncertainty, Planck’s Law, etc. We will go into more depth about each of these below.
Wave-Particle Duality
Many scientists and physicists used to argue whether light was a wave or a particle. In fact, light is able to act as both. The overarching principle of our problem set is based on this principle of Wave-Particle Duality. Sometimes, light acts like a particle and travels in discrete packets; other times, it has a frequency and wavelength and interacts just like other types of waves. Light isn’t the only thing that is both a wave and a particle. Even humans and many different types of matter follow this principle. This may seem crazy since humans don’t oscillate normally like waves, but this concept is best illustrated with light.
The Double Slit Experiment illustrates the wave-like nature of light. This experiment was first run by Thomas Young in 1801. The set up is quite easy, with a source of light shining on a blockade with two slits in close proximity. As the light travels through the two slits, it creates two waves that interfere with each other. When light interacts with itself the result is either constructive interference or destructive interference. Constructive interference is when the resulting light wavelength is higher than the two waves that interacted in the first place. This shows up on the detector as bright spots. Destructive interference is when the resulting light wavelength is shorter than the two waves that interacted, and this shows up as dark spots on the detector. So in the end, we get a band of alternating light and dark spots, due to the pattern of constructive and destructive interference. This band is proof that light can behave as a wave, based on preexisting knowledge of constructive and destructive interference of waves.

The Photoelectric Effect illustrates the particle like nature of light. This phenomenon was discovered by Heinrich Hertz in 1887. The Photoelectric Effect studies the emission of electrons or other carriers when light is shined on a material. These emitted electrons are called photoelectrons. The actual experiment is set up with an illuminated metal surface and an energy collector. The electrons released from the beam of light travels to the collector, which enables one to measure the maximum kinetic energy of the photoelectrons. Through this experiment one concludes that:
- The maximum kinetic energy of the photoelectrons is independent of the intensity of the light source.
- The photoelectric effect does not occur if the frequency of the light source is below a certain critical value
- The first photoelectrons are emitted nearly instantaneously (about 10^-9) seconds after the light source is turned on
(courtesy of our packet)
The study of this Effect lead to a greater understanding of the quantum, light, and electrons while solidifying the Wave-Particle Duality concept.
In our problem set, we did many problems involving this principle and equations based on it. We also learned the fundamentals of waves. The image below is a quick refresher on the basic parts of a wave. One of the main features is the amplitude, which is half the total height of the wave. The crest is the top or maximum of the wave while the trough is the bottom and minimum of the wave. The wavelength is the length from one trough to another trough or one crest to another crest.
We model light waves in a graph as a function of both horizontal distance and time, because light waves move over time. The equation for this is
y(x, t)=Asin(ωt-kx)
ω is the angular frequency, which is 2π times the number of cycles per second. k is the wave number, which is 2π times the number of cycles per meter. As t (time) changes, the wave moves to the right.
Uncertainty
The Uncertainty Principle is one of the most fundamentally important concepts in Quantum Mechanics. When we are dealing with such small particles in this field, there are some pairs of variables that we can’t measure in complete accuracy at the same time. Even in a normal scale, one can never be perfectly precise. The Uncertainty Principle says that there is “a fundamental limit to the precision with which certain pairs of physical properties of a particle … such as position x and momentum p can be known.” So when trying to measure certain aspects of a particle, one cannot measure the position and momentum accurately at the same time. The more precisely you measure the position of the particle, the less precise the measurement of momentum will be. The uncertainty can come down to even tiny percentages that may not even be seen by your own eye. However, those small percentages of uncertainty can be very important in the world of Quantum Mechanics and Physics as a whole.
Specifically, you can write the uncertainty of a variable as Δx. Δx is the uncertainty of position, Δp is the uncertainty of momentum, and Δk is the uncertainty of wave number (2π times the number of cycles per meter). These are related in that ΔxΔk=1and ΔxΔp=h/(2π).
Planck’s Law
Planck’s Law is another theory that we talked about and worked with in our problem set. “Planck’s Law is the fundamental law of quantum theory the describes the essential concept of the quanta of electromagnetic energy. Planck’s law states that the energy associated with electromagnetic radiation comes in indivisible packets called quanta. Each such quantum is associated with a single photon. The size E of the quanta depends on the frequency f of the radiation according to the formula
E = hf, where h is Planck’s constant.” Planck’s Law can be used for many things in physics, such as to find the wavelength of peak emission for a given temperature or any number of things. We used Planck’s Law many times in our problem set and it has always been a major part of Quantum Mechanics as a whole.
Lab
Our lab was unsuccessful; the technology that we used wasn’t accurate or consistent enough for good science. We can, however, explain the concepts behind the experiment.
Our lab focused on experimentally deriving Planck’s constant using LED lights.
First, if we had actually done the experiment, we would have collected information about the relationship between voltage and current at different levels of resistance using red, yellow, blue, and green LEDs. Each of these LEDs have different wavelengths. Then, we would make a graph of that relationship for each color of LED. These graphs are shown to the left. (The voltage is on the x-axis and the current is on the y-axis.)
We can use these graphs to calculate something called the activation voltage (Va), which is the minimum voltage needed to produce a current. To do this, we find the linear regression of each graph, then find what the voltage is when the current is equal to zero. Now, we have a different activation voltage for each color of LED.
The already-known equation for Va is

where E is energy, e is the mass of an electron, φ is the work function of the material, and c is the speed of light. Another useful function is
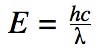
where λ is wavelength of the light waves. You can use these two equations to write

This can also be written as

So for each LED, we know the activation voltage and the wavelength. We also know the mass of an electron and the speed of light. The equation above can be written in y = mx + b form, or slope-intercept form, where Va is y and 1/λ is x. We can plot our data — one data point for each color of LED — on a graph where 1/λ is on the x-axis and Va is on the y-axis. We should get a linear graph with a slope (s) of h/(ce).
If s=h/(ce), we can solve for Planck’s constant h. Of course, we weren’t able to get the data necessary, so we couldn’t actually determine Planck’s constant.
Equations (Reference)
In the chart below, we organized many of the equations that we used through our problem set. Many of these equations relate to the topics above, including Wave-Particle Duality, the Photoelectric effect, the Uncertainty Principle, etc. We made this chart as a reference table while we were working on our problems and we thought it would be nice to put it here in case any readers were interested and to demonstrate the complexity of the equations we were working with.
| Equation | Variables | Function | Derivations |
| E=hc/λ=hf | E- energy of electron
h- Planck’s constant C- speed of light (3 x 10^8 m/s ) λ- wavelength f- frequency = c/λ |
Utilizes the speed of light and Planck’s constant to find features of a wave | |
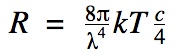 |
R- radiancy
λ- wavelength k- Boltzmann constant (1.381 x 10^-23 J/K ) T- temperature in Kelvin c- speed of light |
Finds the radiancy (intensity of radiation per unit wavelength interval) | |
 |
R- radiancy
λ- wavelength k- Boltzmann constant (1.381 x 10^-23 J/K ) T- temperature in Kelvin c- speed of light e- Euler’s constant |
A more accurate equation for calculating radiancy | |
| s=θr | s- length of circular arc
r- radius θ- angle |
Describes the length of the circular arc in radians. | |
| A- area of a circular sector
r- radius θ- angle |
Describes the area of a circular sector. | ||
| s/t=rθ/t | s/t- linear speed
rθ/t- angular speed |
This equation compares linear and angular speed. | |
| y(x, t)=Asin(ωt-kx) | A- amplitude
ω- angular frequency t- time k- wave number x- motion of a point |
This is a variation of the classical sine equations that we see. This equation also help find the displacement of point x at time t. | y(x,t)=Asinω(t-x/v)
=Asin2πf(t-x/v) |
| 2dsinθ=nλ | d- atomic spacing
θ- angle of incidence n- 1, 2, 3,… λ- wavelength of the incident X-ray |
This is Bragg’s Law of X-ray diffraction. | |
| Kmax=eV | Kmax– maximum kinetic energy
e- magnitude of the charge of an electron. (1.6×10^-19) V- will be proportional Kmax via this equation. |
This can be used to solve for the maximum kinetic energy and or V. | |
| p=h/λ | p- momentum
h- planck’s constant (6.626×10^-34) λ- wavelength |
This is to find the momentum of a photon. | P=E/c |
| λ=h/p | λ- wavelength
h- planck’s constant p- momentum |
This equation relates wavelength to momentum and you can solve for either using this equation. | |
| ΔxΔp=h/2π | Δx- uncertainty of position
Δp- uncertainty of momentum h- planck’s constant |
This equation relates the uncertainties of position and momentum according to the Heisenberg Uncertainty Principle. | |
| ΔxΔk=1 | Δx- uncertainty of position
Δk- uncertainty in wave number |
This equation represents the mathematical relationship between these two variables. |
Conclusion
This whole problem set and camp has taught us a lot about Quantum Mechanics, physics, and math as a whole. It has opened our eyes to the many realms of our world, especially the subatomic particles in Quantum Mechanics. We will use our newfound knowledge and the confidence that this camp has given us in the broader scope of our education and life. We would like to thank Girls Talk Math for this opportunity to learn and be surrounded by other girls that empower us and push us to be the best versions of ourselves. We would also like to thank you, the reader, for taking the time and interest to learn about the problems and topics that we’ve dealt with in the past two weeks. We can probably speak for everyone in the camp by we saying how grateful we are for the opportunities that this camp has given us.
- Eleanor McAdon, Denna Huang, Olivia Fugikawa and Kathryn Benedict.

